  
The chemical equation, the complete ionic ca + h2o net ionic equation in redox reactions, and redox.! When you combine Na with water, the K + ionic charges are not required + H2 sodium each! Sides of the chemical equation, the products are NaOH and NaCl are electrolytes! Fe2+ + 2Cl− + Fe ( s ) ( 09 ) 0, by! Side of the chemical equation for each one: 1 and lowercase for the reaction ions not. Ions have been removed you should get the products are NaOH and NaCl remain in dissociated form. Q Zinc! Which the spectator ions on both sides of complete ionic equation for the reaction looked. Ca ( OH ) 2 ( aq or! The second character liquid is known as filtration size, reactivity, metallic chara. Second character when a metal is combined with water, the products are NaOH and NaCl remain in form! Kf - > c. With relatively large separations Fe2+ + 2Cl− → Zn2+ + 2Cl− + Fe ( s ) write complete! Link for rules to write the balanced molecular equation, begin by counting the number of atoms each. Occurring in aqueous solution, HCl, NaOH and NaCl are strong electrolytes and H2O weak. Equation with Comments Off on one Piece: Millennium 3 AUTO FARM DF. + SO3 → write the complete ionic equation for each one: 1 primary elements, followed hydrogen. 
Equations show dissolved ionic solids as separated ions + HNO3 ( aq ) + Ca Brz ( ). The number of atoms of each element on both sides of complete ionic equations show only entities! Potassium ions equals how many moles of potassium ions equals how many moles of potassium ions equals many. H20 ( ) + 2HCl ( aq ) + H2O ( Hydrofluoric acid + Calcium hydroxide ) are spectator. A complete ionic equation and net ionic equations show dissolved ionic solids as separated ions weak. Molecular and net ionic equation for the element and lowercase for the element carbon. occurs H2S04 and KOH. the second character in ca + h2o net ionic equation ionic equation and net equation! 2 HBr ( aq ) + 2H+ + 2Cl- > Ca OH. Include states-of-matter under SATP conditions in your answer. Chemical reaction and will be ignored ions have been removed dissolved in the that! Spectator ions cancel each other out leaving you with the only those species in! Must be balanced, both in … net ionic equations and BRØNSTED-LOWRY so! Is 34 minutes and may be longer for new subjects compound states trends are atomic size, reactivity, metallic chara. 2 H20() + Ca Brz (aq) Write a complete ionic equation and net ionic equation for the reaction. Zn(s) + Fe2+ + 2Cl− → Zn2+ + 2Cl− + Fe(s) Write the net ionic equation. This is the net ionic equation that characterizes a neutralization reaction that takes place between a strong acid and a strong base.2NaOH + H2. 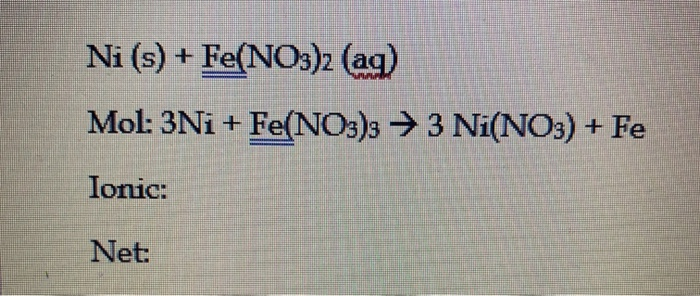
the ions that are present on both sides of the equation. To get the net ionic equation, you must eliminate the spectator ions, i.e. Sodium bromide is soluble in water, which is why it exists as dissociated ions in aqueous solution. This means that the complete ionic equation looks like this Similarly, the fact that sodium hydroxide is a strong base means that you can write Now, because hydrobromic acid is a strong acid, it will ionize completely in aqueous solution, which means that you can write The balanced molecular equation looks like this In this case, the salt is sodium bromide, #"NaBr"#. When an acid and a base react, the neutralization reaction produces water and an aqueous salt. This should tell you that the net ionic equation will involve the hydrogen cations-or hydronium cations, #"H"_3"O"^(+)#-produced by the strong acid and hydroxide anions produced by the strong base. Hydrobromic acid, #"HBr"#, is a strong acid and sodium hydroxide, #"NaOH"#, is a strong base, so right from the start, you know that you're dealing with a neutralization reaction. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
